Pancreatic Ductal Adenocarcinoma
Pancreatic ductal adenocarcinoma (PDA) is lethal cancer with overall survival of six months. Although numerous phase 3 trials have failed to improve overall survival, some patients have responded to drugs used in other malignancies. Although there are no established molecular subtypes of PDA, studies in other solid tumors have revealed that response heterogeneity occurs based on the tumor's molecular profile. By targeting drugs against tumor subtypes, treatment outcomes improve. But despite these promising developments, the scarcity of tumor specimens and the difficulty in identifying molecular subtypes have prevented progress in this area.
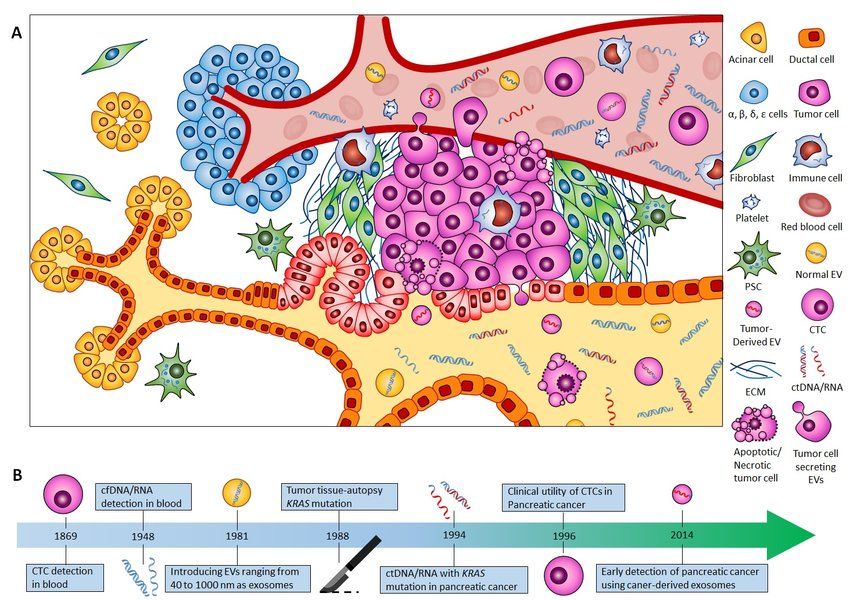
Detection
Detection of ductal obstructive ductal adenocarcinoma (DON) in pancreatic cancer is an essential step toward effective treatment. The disease accounts for 85% to 90% of solid pancreatic neoplasms, and its incidence continues to rise. It is the fourth leading cause of cancer-related deaths worldwide. Imaging plays a vital role in diagnosis and staging and identifying how the disease responds to treatment. Pancreatic neuroendocrine neoplasms have distinct appearances and patterns of spread and should be recognized on imaging to ensure appropriate management.
Currently, no accurate test exists to diagnose PDAC early. However, biomarkers of PDAC have been identified, including sax (a protein found in the blood of asymptomatic patients). The blood-based test has high sensitivity and specificity and has shown good diagnostic value in detecting this disease in advanced cases.
Treatments
Fortunately, there are several effective treatments for invasive ductal adenocarcinomas. Early detection of breast cancer is essential because invasive ductal adenocarcinoma is curable if detected and treated appropriately. Invasive ductal carcinoma can spread to the lymph nodes and other body parts, including the lungs and brain. Patients may feel a lump in their breast, but cancer has spread beyond the milk ducts.
When invasive ductal adenocarcinomas develop, a biopsy of the affected tissue is done. A biopsy is typically done using needles to obtain tissue samples from the breast. Further testing may include ultrasound and MRI scans. Various tests are used to determine the size of the tumor and can help determine whether it is malignant or not. Treatment for invasive ductal adenocarcinoma varies, but the main goal of these treatments is to prevent the development of invasive ductal endocrine cancer.
Prognosis
The prognosis of ductal adenocarcinoma is not good. This aggressive type is frequently found coincident with acinar prostate cancer. However, genomic features driving the evolution toward ductal histology remain unclear. DNA and RNA were extracted from tumor samples to identify molecular markers and their potential clinical applications. Integrated genomic analysis showed that ductal and acinar foci are closely related and may originate from the same cell. However, ductal foci have lower levels of acinar cells.
Diagnosis of ductal adenocarcinoma can be based on immunohistochemical tests or pathological examination. Earlier treatment improves the prognosis of patients. If the tumor is diagnosed early, patients with pancreatic ductal adenocarcinoma will have a five-year survival rate of 20-25%.
Metabolic pathways
In addition to normal cells, tumors change their metabolic profiles. Some types of cancer have different metabolic profiles depending on the type of tissue and genetic lesion responsible for them. A recent study has identified three such metabolic phenotypes in pancreatic cancer. Detailed analysis of these phenotypes can better understand the various types of pancreatic cancers.
Several lipid metabolic pathways are disrupted in pancreatic duct adenocarcinoma. According to a study published in Proceedings of the National Academy of Sciences, a deficiency in a particular pathway can increase the risk of developing the disease. Mutations of the mTOR gene, ArgA1, and KRAS are also associated with increased tumor-causing potential.




