Endosonographie and Olaparib-Erhaltungstherapie for Pancreatic Cancer
Pancreatic cancer is a disease affecting the pancreas, which is often the cause of diabetes. This disease is diagnosed through various diagnostic procedures, including sonography, computer tomography, and Kernspintomography. The extent of tumor spread and metastatic disease determine operability. Treatment options range from radiation therapy to surgery, and endoscopic procedures include Endosonographie and Olaparib-Erhaltungstherapie.
Exocrine
Patients who receive the chemotherapy drug Exokrin for pancreatic cancer can expect their disease to shrink significantly. Despite the potential benefits, many patients are unsure of the drug's long-term side effects. The tumor may even return if the treatment is not practical. Pancreatic cancer can produce within the pancreas or in distant locations, similar to metastasis.
Pankreaskarzinome
PSP Pankreaskarzinome is an initiative to stimulate clinically relevant research on pancreatic cancer. The industry brings patients, family members, doctors, nurses, and researchers together. This research project is based at the Surgical University Clinic in Heidelberg and is supported by the Medical Faculty of the University of Heidelberg ethics committee. Its objectives include determining the optimal treatment and establishing a global registry to track pancreatic cancer treatments.
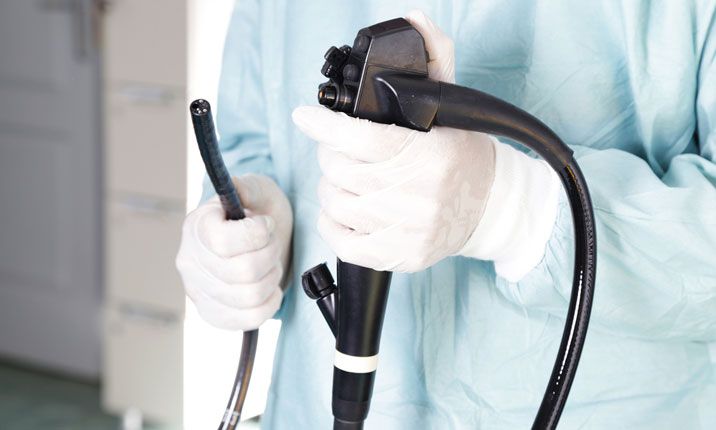
Endosonographie
An Endosonographie for pancreatic cancer involves using ultrasound waves to take a picture of the pancreas. This test is more accurate than a CT scan, the standard pancreatic cancer diagnosis. The technique involves inserting a thin, lighted tube into the patient's mouth, stomach, and the first part of the small intestine. The endoscope is then inserted through the abdomen. Ultrasound waves bounce off of organs, and their patterns can be used to identify any small tumors. X-rays and ultrasound techniques are also used to guide the endoscope. Then, the technician uses the endoscope to remove pancreatic cancer cells.
Olaparib-Erhaltungstherapie
A significant randomized phase III trial of Olaparib for the treatment of advanced pancreatic cancer reported encouraging results. It increased the median progression-free survival time, previously three to seven months, to almost four months. Further, more than twice as many patients were tumor-free after one year or two years. Although this benefit has not been proven, the European Commission recently approved expanded approval of Olaparib for pancreatic cancer. The European Arzneimittelbehorden accepts the trial results as long as it results in progression-free survival.
Thrombose
Thrombose in the pancreas is a relatively common complication of the disease. Thrombose in pancreatic cancer is associated with an intrinsic hypercoagulable state, in which the tumor cells activate platelets and express procoagulant factors. This activation of coagulation is thought to be related to the tumor's enhanced growth and angiogenesis. Thrombose in pancreatic cancer can manifest as either arterial or deep venous thrombosis. It is estimated that between one to five percent of patients with pancreatic cancer will develop thrombosis. Treatment options include anticoagulation with warfarin.
New Pigmentations of the Haut
The most common symptoms of pancreatic cancer are a yellowish skin tone, which may appear as squamous cells. Although the disease is generally curable, some tumors may spread to other body areas, including lymph nodes and the stomach. In such cases, some surgical treatments are recommended. However, pancreatic cancer may also produce signs and symptoms that are not immediately noticeable, such as the yellowing of the eyes and skin.
Treatment
While standard drug therapy for end-stage pancreatic cancer doesn't work very well, German hospitals offer more advanced treatments that improve patient outcomes. Proton therapy and targeted therapies are two of these options. Both methods are used to kill cancer cells without damaging healthy tissue. Patients with advanced pancreatic cancer experience unpleasant side effects, such as pain, nausea, vomiting, diarrhea, and weakness. Some patients may require surgery to eliminate these symptoms.




